Advancing candidates designed to address resistance mechanisms in multiple cancers
Our portfolio of novel agents targets multiple resistance mechanisms by leveraging our specialized expertise in the following areas:
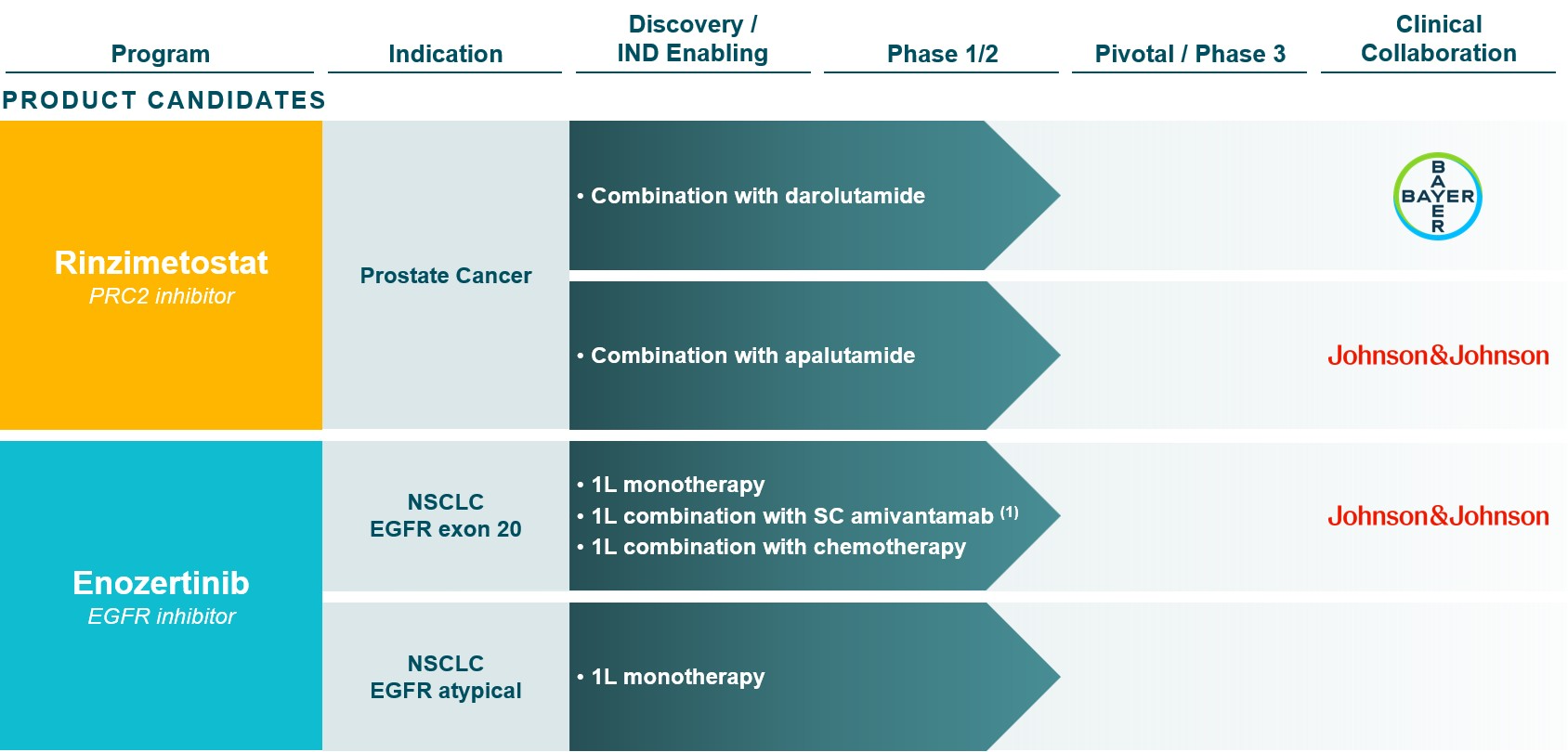
PRC2 INHIBITOR PROGRAM: Rinzimetostat
The dysregulation of PRC2 methyltransferase activity is found in a wide range of cancers including prostate cancer, lung cancer, colorectal cancer, breast cancer, and hematological malignancies. PRC2 is composed of two druggable subunits: EED and EZH2. Several companies are developing EZH2 inhibitors; however, the pharmacologic properties of these compounds result in high doses that may achieve only partial target inhibition in the clinic. Additionally, preclinical studies suggest drug resistance to EZH2 inhibitors may develop via EZH1 bypass compensation or acquired mutations in EZH2, thus allosteric inhibition of PRC2 via EED may have benefits over EZH2-mediated inhibition of PRC2. Rinzimetostat is a potent and selective allosteric inhibitor of PRC2 via the EED subunit that was designed to have superior drug properties over EZH2 inhibitors and is efficacious in androgen receptor pathway inhibitor (ARPI) -sensitive and -resistant prostate cancer models in preclinical studies. We filed and cleared an IND with the FDA for rinzimetostat in 2021.
In 2024, we reported initial data from the Phase 1b trial of rinzimetostat as a single agent in patients with advanced prostate cancer, demonstrating potential best-in-class drug properties, including long clinical half-life, robust target engagement, and a favorable safety profile.
In November 2025, we reported Phase 1b dose exploration data in 20 mCRPC patients treated with rinzimetostat in combination with apalutamide or with darolutamide. Data demonstrated PSA responses and ctDNA reductions across all rinzimetostat dose levels and at comparable rates in combination with apalutamide or with darolutamide. Both combination regimens demonstrated a safety profile compatible with long-term dosing, with the vast majority of treatment-related adverse events (TRAEs) Grade 1 or 2 in severity and consistent with PRC2 and AR inhibition. Only one patient experienced a Grade 3 TRAE, and there were no Grade 4 or Grade 5 AEs attributed to rinzimetostat, apalutamide or darolutamide.
Also in November 2025, we announced the completion of the dose exploration portion of the Phase 1b trial and the selection of provisional recommended Phase 2 doses (RP2Ds) of rinzimetostat to be tested in combination with darolutamide and apalutamide in the dose optimization portion of the Phase 1b trial.
In March 2026, we reported Phase 1b dose optimization data in mCRPC patients previously treated with abiraterone and the selection of 400 mg once daily rinzimetostat in combination with darolutamide as the recommended Phase 3 dose (RP3D). At the selected RP3D, rinzimetostat in combination with darolutamide demonstrated data supporting its potential best-in-disease profile. At a median follow-up of ~5 months, the landmark 5-month rPFS of 84% was consistent with a competitor PRC2 inhibitor benchmark, and better than the benchmarks for standard of care therapies in mCRPC. Importantly, data demonstrated a highly differentiated, potential best-in-disease safety profile, with significantly lower frequency and severity of adverse events, nearly all Grade 1 or 2, and far fewer treatment modifications than competitor regimens. We expect to initiate our first global Phase 3 registrational trial for rinzimetostat, named Himalayas-1, in post-abiraterone mCRPC patients.
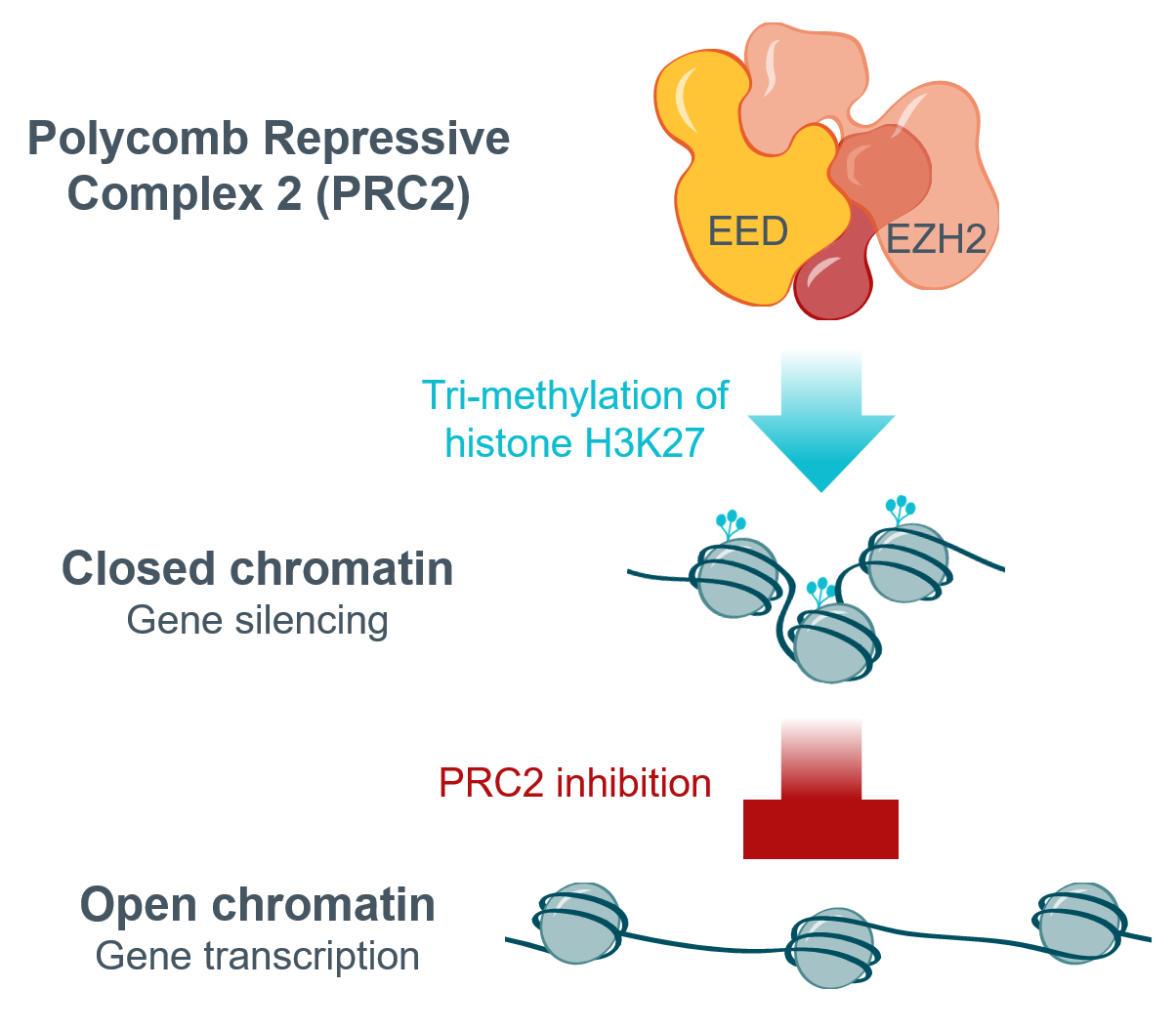
- Two druggable subunits:
– EED: responsible for histone binding; target of rinzimetostat
– EZH2: responsible for histone methylation; target of first-generation inhibitors - Dysregulation of PRC2 linked to several cancers
– Decreased expression of target genes associated with poor prognosis in prostate cancer - First-generation inhibitors, designed to inhibit EZH2, have demonstrated promising clinical activity and chemoresistance
therapeutic potential in prostate cancer, among other indications
Brain-penetrant EGFR inhibitor program: Enozertinib
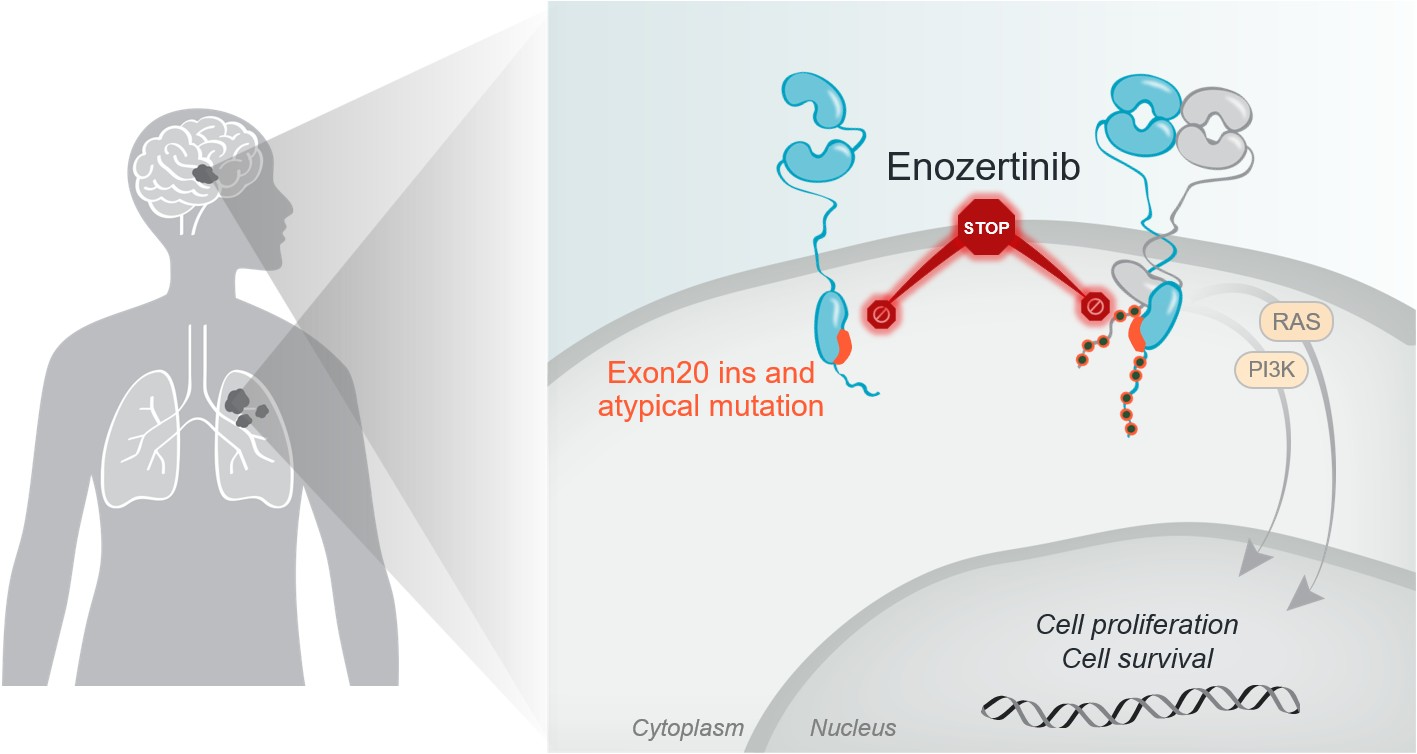
The ErbB receptor tyrosine kinase family is involved in key cellular functions, including cell growth and survival. Epidermal Growth Factor Receptor (EGFR) mutations are observed across multiple solid tumors, including NSCLC, breast, gastrointestinal, bladder and other cancers. EGFR exon 20 insertion mutations and EGFR P-loop and alpha C-helix compressing (PACC) mutations – a key subset of atypical mutations – are collectively observed in approximately 5% of all patients with NSCLC in the US. Approximately 30% of patients with EGFR mutant NSCLC have known CNS disease at diagnosis, and approximately 50% develop brain metastases over the course of their disease, with the brain often the first site of progression, which contributes to poor prognosis.
Enozertinib is a brain-penetrant, orally bioavailable, irreversible inhibitor designed to selectively target EGFR with high potency against exon 20 insertion and atypical mutations. Enozertinib has demonstrated greater brain exposure in preclinical studies compared to certain other compounds being developed against exon 20 mutations and has shown strong anti-tumor activity in an EGFR-driven intracranial lung cancer model.
The CTA for enozertinib in South Korea was cleared in the first quarter of 2022. We also filed and cleared an IND with the FDA for enozertinib in the third quarter of 2022.
Enozertinib is being evaluated in Phase 1b trials in EGFR exon 20 insertion and EGFR atypical mutated NSCLC. The trials allow enrollment of patients with CNS metastases that are either treated or untreated but asymptomatic. We reported initial Phase 1b data with enozertinib as a monotherapy in patients with EGFR exon 20 insertion mutations at the 2023 ESMO Congress, which demonstrated both systemic and intracranial activity in a heavily pre-treated patient population across multiple dose levels. We reported additional Phase 1b data at the 2025 ESMO Asia Congress in treatment-naïve and in previously treated NSCLC patients with EGFR exon 20 insertion and EGFR PACC mutations. Enozertinib achieved highly competitive systemic response rates as well as profound antitumor activity in the CNS. Enozertinib also demonstrated a well-tolerated safety profile, with no significant off-target toxicity and manageable on-target toxicity, resulting in a low rate of discontinuations. Based on these data, 80 mg once daily oral enozertinib has been selected as the monotherapy dose for potential Phase 3 development. Dosing and follow-up continues in NSCLC patients with exon 20 insertion mutations, including as a monotherapy, in combination with amivantamab and hyaluronidase-lpuj subcutaneous injection (SC amivantamab) and in combination with chemotherapy, as well as in NSCLC patients with EGFR atypical mutations as a monotherapy.

 OricPharma logo
OricPharma logo